OpenClinica Enterprise EDC
Products
Information
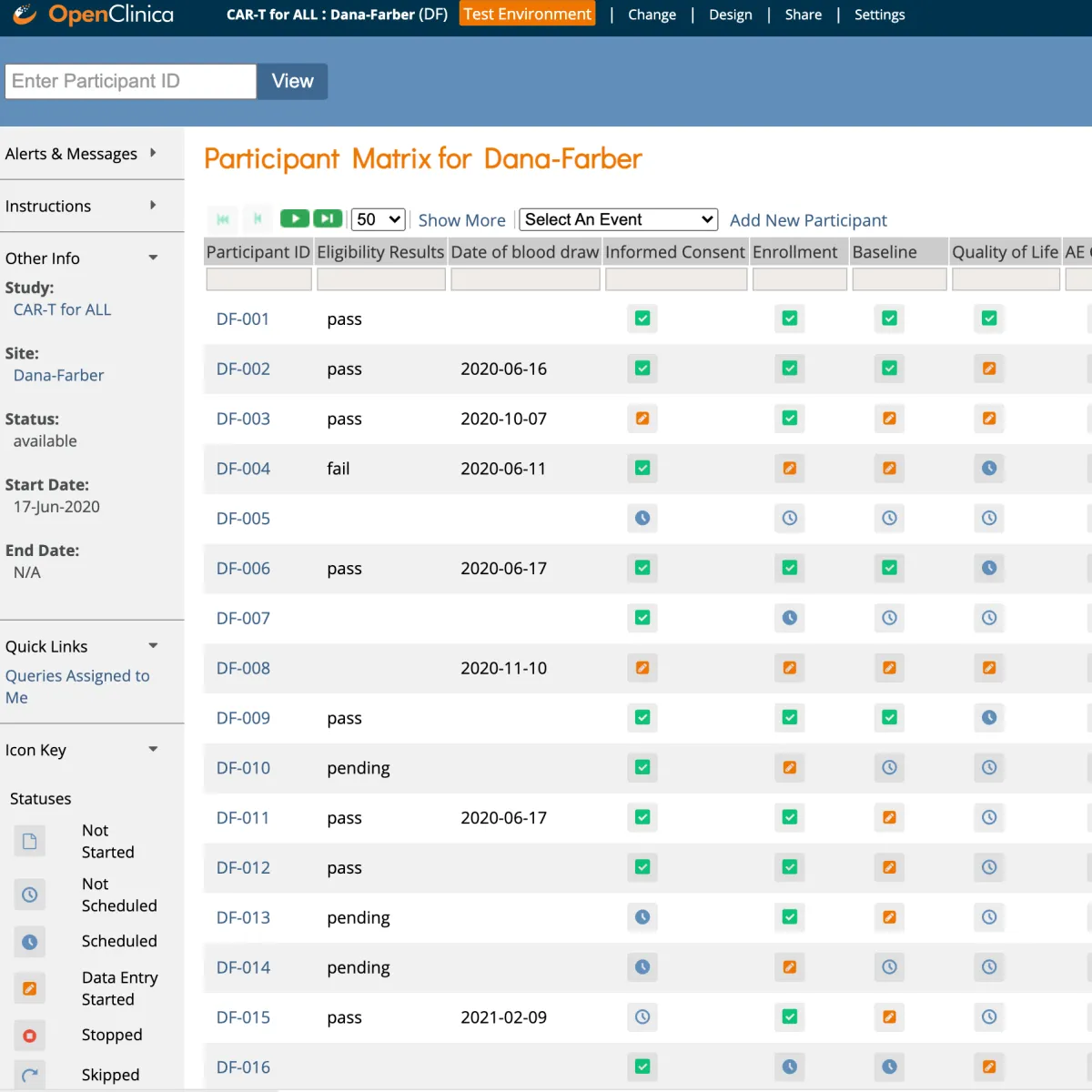
OpenClinica simplifies the capture, cleaning, and management of clinical research data. Our SaaS EDC solution is trusted and proven in over 10,000 studies for the past 15 years. OpenClinica has a global customer base, subject to regulations including GCP, 21 CFR Part 11, HIPAA, and GDPR, and we are regularly audited by our customers to ensure compliance. We support RCTs, registries, platform trials, and many other research designs. OpenClinica helps you capture better data, faster by:
- Being built around richly interactive, mobile-friendly eCRFs that execute logic in real-time, with layout control as simple as Word
- Increasing efficiency at study startup, with intuitive drag-and-drop study setup tools and content assets that can be reused and customized
- Combining the study build environment with single-click validated design->test->production pathways to ensure version control and data integrity for your studies and amendments
- Accelerating study timelines by making it easy to capture and track complete, high-quality research data from diverse sources. Our data management & monitoring feature sets include intelligent query management, protocol versioning (including site-specific customizations), SDV, forced reason for change, and more.
- Providing easy, self-service onboarding for all user types, with the ability to define custom roles with associated access privileges
- Ensuring seamless integration through an extensive RESTful web services API
- Enabling on-demand access to data via export (Excel, SAS, SPSS, HTML, and CDISC ODM XML)
- Ensuring your compliance with audit trails, electronic signature capabilities, security, a robust quality system, and meeting GCP, 21 CFR Part 11, HIPAA, and GDPR requirements
- Delivering a world-class infrastructure and information security - SSL encryption, encryption at rest, data center security certifications: SSAE16 SOC1, SOC2 Type II, ISO 27001, trusted reliability with 99.99% uptime, 24/7/365 system monitoring
